Invectys anti-HLA-G CAR-T cells demonstrate their anti-tumor efficacy in vivo
These results open the way to targeting tumors via HLA-G, a powerful immune checkpoint and a cancer-specific antigen.
Paris, February 4, 2020 – Invectys today announces positive in vivo results for its anti-HLA-G CAR-T cells. Invectys has identified the HLA-G molecule as a high-potential tumor target, particularly in solid tumors. HLA-G is mainly involved in pregnancy, where it prevents the destruction of the fetus by the mother’s immune system.
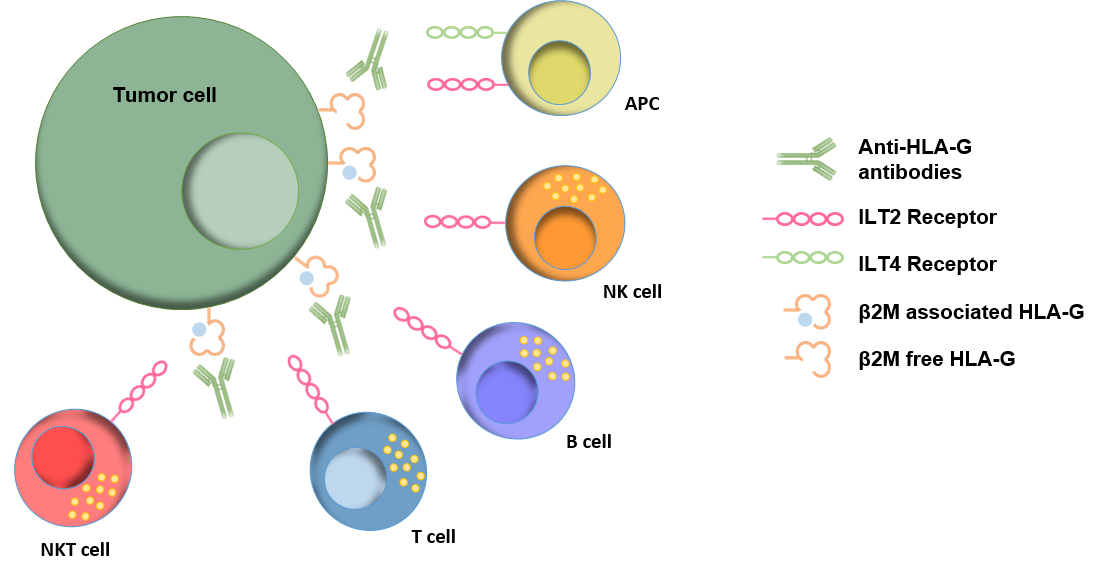
HLA-G has recently been identified as an immune checkpoint (in English Immune Checkpoint, or ICP) generating a tumor microenvironment (in English Tumor Microenvironment, or TME) to counter the host’s immune response. HLA-G is present on tumor cells in TME, but almost absent from healthy tissues, which makes it a more specific and more prevalent tumor antigen than PD-L1.
CAR-T therapies have demonstrated their potential, in particular in B-cell malignancies. However, their use in solid tumors remains difficult. The main obstacles are: few highly specific antigens, low penetration of T cells into tumors, and the immunosuppressive MTCT often associated with these tumors which weakens the immune response.
New specific antibodies
The Invectys Discovery team generated anti-HLA-G antibodies, which enabled the development of 3rd generation anti-HLA-G CAR-T. Anti-HLA-G CAR-T are insensitive to the ICP effect of HLA-G, and have demonstrated their specific cytotoxicity against tumor cells expressing HLA-G, giving them a long-term memory phenotype and a strong anti- tumor in vivo, controlling tumor progression for up to 60 days.
Target solid tumors with anti-HLA-G CAR-T cells
These anti-HLA-G CAR-T should not only destroy solid HLA-G + tumors, but also disturb the associated TME. Indeed, by targeting HLA-G, on the one hand the carrier cells are destroyed, and on the other hand the CAR-T neutralize the immunosuppressive TME induced by HLA-G which protects the tumor from the host’s immune reaction.
The CAR-T anti-HLA-G project was awarded the “Best Project” 2019 by the MATWIN International Board and a “Merit award” from the ESMO Immuno-Oncology Congress 2019.