Cusabio Helicobacter pylori Recombinant
Bottom
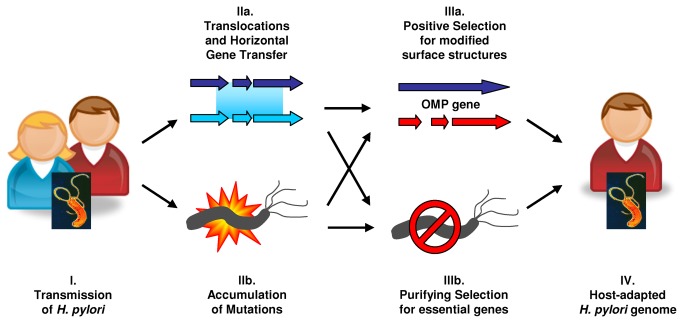
Helicobacter pylori Recombinant is a human pathogen, and during the infection process, antigens from the bacterium elicit strong humoral immune responses in the host. In our previous report, the native H. pylori protein UreG showed good reactivity with sera from H. pylori patients. This study aimed to produce the recombinant form of the protein (red) and determine its seroreactivities.
Methods
The H. pylori UreG coding sequence was cloned and the recombinant protein was expressed and purified by affinity chromatography using nickel nitrilotriacetic acid (Ni-NTA) resin. The antigenicity of rUreG to detect H. pylori-specific antibodies was determined by Western blotting, using HRP-conjugated anti-human IgG and IgA antibodies as probes. A total of 70 sera were used, comprising 30 positive and 40 control serum samples. The positive sera came from culture-positive H. pylori-infected patients with duodenal ulcers, gastric ulcers, or gastritis.
Control sera comprised three types of samples with no detectable H. pylori antibodies, ie, healthy individuals (no history of gastric disorders) (n = 10); patients who attended an endoscopy clinic (because of gastrointestinal complaints) but whose H. pylori culture was negative (n = 20); and people with other diseases (n = 10). In addition, serum from hyperimmune mice against rUreG was raised and tested with native and recombinant UreG protein.
Keywords: Helicobacter pylori, UreG, Cloning, Expression, Recombinant protein, Seroreactivity
Purity: greater than 85% as determined by SDS-PAGE.
Destination names: ftnA
Uniprot No.: P52093
Research Area: Others
Alternative Names: ftnA; pfr; HP_0653Non-heme bacterial ferritin; EC 1.16.3.2
Species: Helicobacter pylori (ATCC strain 700392/26695) (Campylobacter pylori)
Source: Baculovirus
Expression Region: 1-167aa
Mole Weight: 21.8 kDa
Protein Length: Total length
Tag Information: N-terminal 10xHis-tagged
Form: Liquid or Lyophilized Powder
Note: We will preferably ship the format we have in stock, however, if you have any special requirements for the format, please remark your requirement when placing the order, we will prepare according to your demand.
Buffer
If the dosage form is liquid, the default storage buffer is Tris/PBS based buffer, 5%-50% glycerol.
Note: If you have any special requirements for glycerol content, please remark it when you order.
If the administration form is a lyophilized powder, the buffer before lyophilization is a Tris/PBS-based buffer, 6% trehalose, pH 8.0.
Reconstitution
We recommend that this vial be briefly centrifuged before opening to bring the contents to the bottom. Reconstitute protein in sterile deionized water at a concentration of 0.1-1.0 mg/mL. We recommend adding 5-50% glycerol (final concentration) and an aliquot for long-term storage at -20°C/-80°C. Our final default glycerol concentration is 50%. Customers could use it for reference.
Storage Conditions
Store at -20°C/-80°C upon receipt, need to be aliquoted for multiple uses. Avoid repeated cycles of freezing and thawing.
Shelf life
Shelf life is related to many factors, storage condition, buffer ingredients, storage temperature and the stability of the protein itself. Generally, the shelf life of the liquid form is 6 months at -20°C/-80°C. The shelf life of the lyophilized form is 12 months at -20°C/-80°C.
Delivery time: 3-7 business days
Notes: Repeated freezing and thawing is not recommended. Store working aliquots at 4°C for up to one week.
Results
The urea gene fragment was successfully cloned and expressed in both soluble and insoluble forms. Western blots on rUreG protein showed 70% (21/30) and 60% (18/30) reactivity with patient sera when challenged with HRP-conjugated anti-human IgG and IgA antibodies, respectively; and the combined IgG and IgA western blots showed 83.3% (25/30) reactivity.
In comparison, 97.5% and 92.5% of the control sera showed no reactivity when tested with HRP-conjugated anti-human IgG and IgA antibodies, respectively. Both H. pylori lysate antigen and rUreG protein showed a distinctive band at the expected molecular weight when tested with serum from hyperimmune mice.
Conclusion
The red protein was successfully cloned and expressed and showed good reactivity with H. pylori culture-positive patient sera and no reactivity with most control sera. Therefore, the diagnostic potential of this recombinant protein deserves further investigation.